![]() As outlined in this study, silicone rubber has a range of impressive qualities for innovative uses in a variety of industries.
As outlined in this study, silicone rubber has a range of impressive qualities for innovative uses in a variety of industries.
The main element in silicone is silicon (Si), which is the second most abundant element in the earth’s crust after oxygen.
In nature, silicon has a strong affinity for oxygen (SiO₂). At temperatures over 1900 °C, it is reduced to metallic silicon, which reacts with other compounds to produce silicones, a class of synthetic resins with both organic and inorganic characteristics. Silicones consist of a silicon–oxygen backbone chain known as a siloxane bond – Si – O – Si – O – with two organic groups attached to each silicon centre – (-SiR₂-O-)n-.![]()
These highly functional elastomers have both organic and inorganic characteristics. The different polymer side groups and structures give the materials specific combinations of properties, as well as giving the various subclasses of silicones their names.
Names according to ASTM D1418 and ISO1629:
MQ (polydimethylsiloxane)
VMQ (polyvinylmethylsiloxane)
PVMQ (polyphenylvinylmethylsiloxane)
FVMQ (polyfluorovinylmethylsiloxane).
The siloxane bonds (Si-O-Si) that form the backbone of the silicone chains are highly stable and their bond energy (433KJ • mol¯¹) is higher than the carbon–carbon bonds (348KJ • mol¯¹) and carbon–oxygen (358KJ • mol¯¹) that make up the structure of organic rubbers, and silicone rubbers therefore have greater chemical, thermal and environmental stability. Silicone molecules tend to take on a 360° helical structure, which results in impressive flexibility and elasticity even at very low temperatures. The methyl groups along the molecular chains give the material powerful hydrophobic and release properties; these properties are largely unaffected by temperature. HTV silicone polymers can be crosslinked with two different curing agents: peroxide and platinum.
Peroxide crosslinking takes place through the breakdown of organic peroxides into free radicals, which attach themselves to the organic groups present in the polymer, forming new C – C bonds as shown in the following diagram:
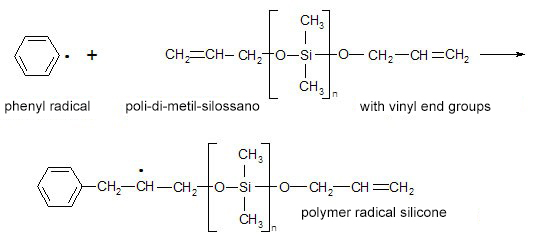
Finally, the free radical silicone polymers recombine with each other to form a crosslinked polymer.
The crosslinking catalysed by the platinum complex is a polyaddition process very similar to that of polymerisation. The platinum catalyst causes the vinyl double bonds of the side chains to create a bond with the Si-H groups on the polymethylhydrodimethylsiloxane chains.
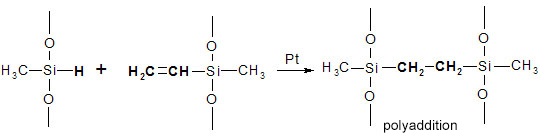
As shown in this diagram, there are no by-products. The working environment is therefore free from vulcanisation fumes, and the moulded material has improved physical/mechanical properties: better tear resistance, greater elongation at break, and high transparency with no yellowing.
This brief introduction to the chemistry of silicones should help you understand their unique characteristics, as described below.
 Temperature resistance
Temperature resistance
The operating temperature of silicone is between -110 °C and + 320 °C. More precisely, silicone rubber can be used for thousands of hours at 200 °C without any effect on its properties, while specially formulated materials can withstand temperatures up to 320 °C for defined periods. Similarly, the embrittlement of silicone occurs at temperatures below -60 °C, while phenyl silicones (PVMQ) can withstand usage temperatures of -110 °C.
Resistance to atmospheric agents
The properties of silicones are not affected by extreme heat, frost, humidity or aridity. They are also extremely resistant to UV rays and ozone. A classic example is the ozone produced by electric motors, which deteriorates most organic rubbers.
Resistance to water and steam
Silicone can be immersed in water from 0 to 100 °C for very long periods without any effect on its physical and mechanical properties. Its low crosslinking density, however, makes it permeable to most gases; silicone can withstand steam for short periods at temperatures no higher than 120 °C, as under these conditions the Si-O bonds begin to break down through hydrolysis, leading to the deterioration of the material’s physical/mechanical properties. Special polymers with high thermal resistance offer improved performance under these conditions. For the aforementioned reasons, silicone is not ideal for applications that involve rapid decompression, for example.
Resistance to oils and solvents
Despite having an average resistance to oils at room temperature, silicone cannot match the performance of NBR. However, when the usage temperature rises above 100 °C, organic rubbers begin to lose their properties, while silicones remain stable. Certain types of silicone polymers have also been enhanced in this regard, making them ideal for products that come into contact with mineral oils.
The nonpolar nature of silicones ensures excellent resistance to all polar solvents, diluted acids and bases. On the other hand, the lower the molecular weight of the solvent, the more they tend to swell in contact with hydrocarbons; this phenomenon is totally reversible because silicones do not dissolve. In contact with oils, fuels and chemical agents—where traditional silicones would undergo marked deterioration—fluorosilicone (FVMQ) offers exceptional performance from -70 to 230 °C. Fluorosilicone is a top-of-the-range silicone product, costing up to 10 times more than normal silicone; it is therefore used in fields where its chemical resistance and physical properties are required at a wide range of temperatures.
Thermal conductivity
As an insulating material, silicone does not have good thermal conductivity: at 25 °C, it is 0.2W m¯¹K¯¹.
Special fillers can be incorporated into the silicone mould to reach thermal conductivity values of up to 2.3W m¯¹K¯¹.
Electrical properties
Silicone rubber is an ideal insulating material (1TΩ•m-100TΩ•m), maintaining these properties over a wide range of usage temperatures. Silicone is therefore widely used as an insulator for electrical cable coverings and high voltage systems, replacing epoxy resins as a high voltage insulator (ASTM method arc resistance over 180 sec) and providing better flame resistance, opacity and smoke toxicity.
By introducing different quantities of carbon black, we can create conductive silicones—with resistivity from 0.01 to 10Ω•m—without affecting the mechanical properties of the silicone rubber. These materials are used, for example, for the contact points of keyboards, shielding high voltage cables, and moulding electrodes for electrostimulators.
Flame resistance
Fire-resistant materials are classified through flame propagation and smoke toxicity and opacity tests. Silicone rubber does not catch fire easily, but once the fire has started it will continue to spread without releasing asphyxiants or toxic fumes. It can also be made self-extinguishing by adding small amounts of metal hydroxides, allowing us to obtain compounds that comply with the UL94 V0 or NF F16-101 L2 F1 standards.
Attachment to metal/plastic
When correctly formulated, silicone rubber can be used to produce insert moulded items in combination with metals, plastics and other elastomers that have been prepared and treated with a specific adhesion primer.
Physiological inertness
Thanks to its inalterability and physiological inertness, silicone is suitable for direct contact with food, skin, medicines and drinking water, in line with the main regulations in these areas (FDA 177.2600, BFR BGVV, NSF, WRAS, WRC, KTV, DM2103/73 and EC directive 2002/72). It is also suitable for use in controlled environments, as it does not support microbiological growth. Medical grade materials are produced with special measures to create catheters, prostheses, machine components and other medical devices that comply with the USPal.VI and ISO10993-1 standards.
Aesthetic properties
In terms of elastomers, silicone is the ideal material to produce designer items with high aesthetic standards. Thanks to its transparency, silicone can be coloured in an infinite range of shades, including fluorescent, phosphorescent, metallic, pearlescent, iridescent, and thermosensitive designs. Its ductility makes it suitable for moulding complex shapes, while its tactile qualities, lack of efflorescence, and aesthetic inalterability allow us to create harmonious and stable items.
Environmental factors
Last but not least, we will examine silicone’s environmental impact.
A study conducted by an independent body showed that replacing common goods derived from organic materials with silicone items allows for greater efficiency with regard to the consumption of energy and raw materials, with a positive CO₂ balance and enormous benefits in terms of greenhouse gas emissions around the world.
For more information, you can read the study by visiting the www.siliconescarbonbalance.com website.
Conclusion
Hygienically safe enough to be used for bottle teats and strong enough to withstand the stresses of a shuttle engine, silicone rubber is a truly extraordinary material.
Its unique combination of chemical and mechanical properties mean that this highly versatile polymer is ideally suited to any product market.
By applying our scientific knowledge and technical skills, our company is able to produce perfectly functional and aesthetically flawless silicone items. Our experience in the field of silicone moulding allows us to provide our customers with reliable and durable solutions, creating lasting partnerships in a variety of fields. Since our company was founded, we have created hundreds of different custom moulded silicone items, such as:
- flat and shaped gaskets for lighting
- designer lighting technology pieces
- suction cups with or without stainless steel inserts
- bellows with or without stainless steel inserts
- plates for electrostimulators
- insulators
- membranes with or without inserts
- anti-vibration mounts
- clothing accessories
- boating accessories
- oven gaskets
- cookware gaskets and accessories
- gaskets and accessories for hobs
- trivets
- sterilisation mats for surgical instruments
- dental equipment accessories
- eyewear accessories
- juggling accessories
- fitness accessories
- balls
- valves
- toys
- caps
- keyboard covers
- switch covers
All of our products are manufactured in accordance with the customers’ specifications, using specially formulated and coloured compounds to suit each moulded item.



